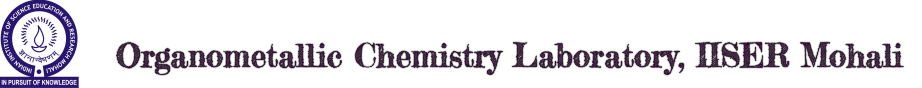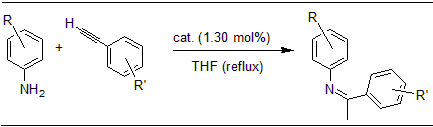Major Research Interests
Organometallic Chemistry of Main Group and Transition Elements
Organometallic Chemistry of Main Group Elements, Borenium Ions and Cationic Organo-aluminum Complexes: Chemistry of highly reactive organo-main group ionic compounds supported by bulky iminophosphonamide ligands with N2P or N3P2 skeleton is the major emphasis of this research. Recently discovered series of cationic hydroborenium ions and their aluminum congeners have been isolated with weakly coordinating anions. These cationic complexes exhibit very strong Lewis acid character and have been useful in promoting/catalyzing organic reactions mediated by Lewis acids.
For details see: Inorg. Chem. 2020, 59, 7195−7203; ChemCatChem, 2020, 12, 2407–2411; Chem. Eur. J. 2018, 24, 4794-4799; ChemistrySelect, 2017, 2, 2039-2043; ChemistrySelect, 2016, 1, 3601-3606; Dalton Trans. 2014, 43, 16880-16888.
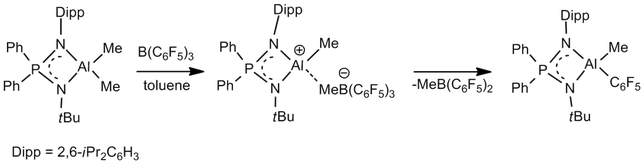
Fine tuning of Lewis acidity of hydridoborenium cations with weakly coordinating bulky anions: We have been successful in the synthesis of hydridoborenium cations with weakly coordinating bulky anions such as [B(C6F5)4]- or [HB(C6F5)3]-. We have also elegantly demonstrated how one can fine tune the Lewis acidity of cationic boron species again with the case of borenium complexes supported by bis(phosphinimino)amide ligands. As shown below the DMAP adduct of the borenium species exhibits its Lewis acid behavior.
For details see: Chem.-Eur. J. 2016, 22, 11035-11041; Eur. J. Inorg. Chem. 2015, 2565-2673.
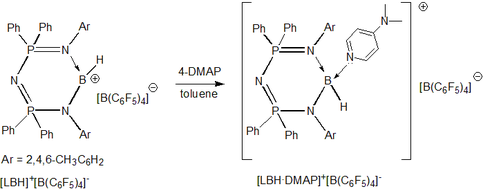
Hydroborenium cations without weakly coordinating anions: We have also discovered that if the supporting ligand is capable of offering kinetic stability to the borenium cations then the use of weakly coordinating bulky anions may not be necessary. This fact has been clearly demonstrated in the preparation of hydroborenium ion with HBCl3- as the counter ion.
For details see: Chem.-Eur. J. 2016, 22, 11035-11041.
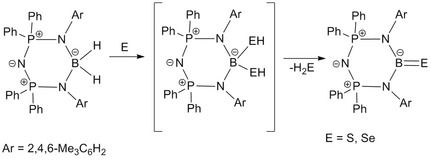
Preparation of boron-chalcogen compounds: The N3P2 backbone of bis(phosphinimino)amide stabilized dihydroboron species under oxidative insertion of S and Se with concomitant release of H2S and H2Se respectively, afforded the multiply bonded terminal boron-chalcogen complexes in the form of terminal thioxo- and selenoxo-borane compounds.
For details see: Dalton. Trans. 2015, 44, 15779-15785.
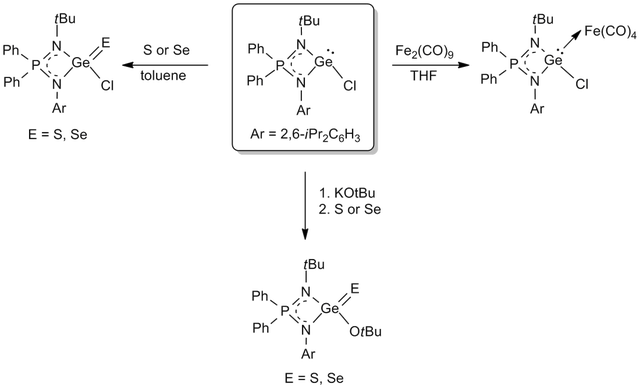
Low Valent Complexes of Group 14 Elements: Synthesis of novel germylene, stannylene chelate compounds stabilized by iminophosphonamide and bis(phosphinimino)amide ligands is under investigation. The successful synthesis of Ge(II) and Sn(II) compounds has opened up a new opportunity to explore Lewis acid-base chemistry, substitution reactions, reduction of these divalent species (to prepare zero valent species) and oxidation reactions (to new interesting derivatives of the unknown tetravalent metal centers). Synthesis of germaacid chloride and germaesters of heavier chalcogens (S and Se) are shown as examples.
For details see: Dalton Trans. 2016, 45, 6079-6087.
Chemistry with Carbene-Transition Metal Complexes
(1) Cyclic(Alkyl)(Amino)(Carbene) Complexes with Late Transition Metals and Catalysis
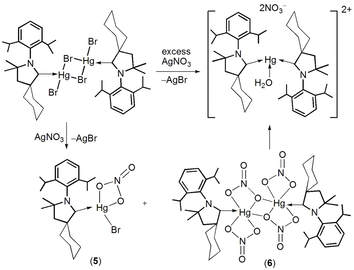
Our efforts to stabilize late transition metal ions with cyclic(alkyl)(amino)(carbene) (cAAC) resulted into the first synthesis of neutral (cAAC) adducts of halide bridged dimers Hg(II); [cAACMe·HgX(μ-X)]2, [cAACcy·HgX(μ-X)]2 (X = Cl, Br, I), and nitrate complexes; cAACcy∙HgBr(NO3) and [cAACcy∙Hg(NO3)(µ-NO3)]2. Some of these complexes were further exploited to prepare cationic Hg(II) complexes such as [(cAACMe)2Hg(NO3)]+[NO3]‒ and [(cAACCy)2Hg]2+[Hg2Cl6]2‒.
Adduct [cAACcy·HgBr(μ-Br)]2 catalyzes intermolecular hydroamination of aromatic alkynes and amines under mild conditions to afford aromatic imines. The reactions proceed under mild conditions and good yields of the products were obtained.
For details see: Dalton Trans. 2018, 47, 6274-6278; J. Organomet. Chem. 2019, 880, 108-115.
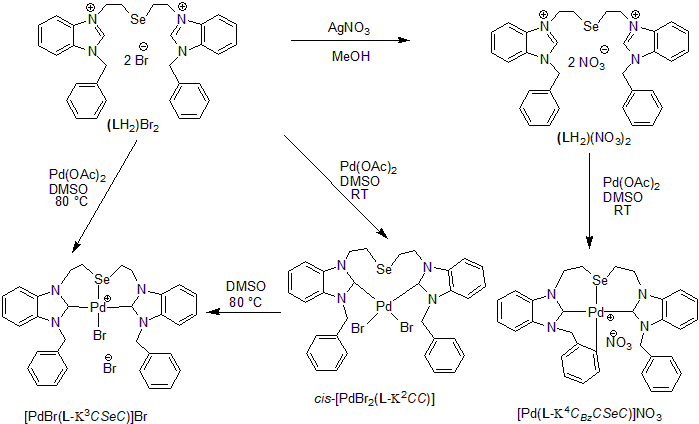
(2) N-heterocyclic/CAAC/BICAAC Carbene Complexes of Transition Elements for Catalysis: We have synthesized novel pincer type/tripodal N-heterocyclic carbenes bearing chalcogens in their backbone. Transition metal complexes of these carbenes are being investigated for their structure and reactivity. The example below illustrates how different salts of the same carbene precursor, under slightly different reaction conditions, can afford metal complexes of different structure and bonding. Recently, we have also discovered that Au(III) and Pd(II) complexes prepared in this project show excellent activity for C-H activation in aromatic rings and promising C-C coupling reactions.
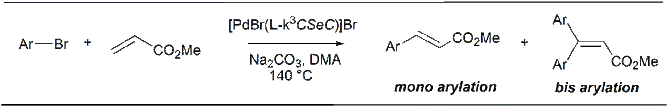
The pincer complex of selenoether bridged carbene with palladium(II)bromide showed very good catalytic behavior for conventional mono- as well as rare bis-arylation of deactivated esters in the presence of arylbromides.
For details see: Dalton Trans. 2017, 46, 6291-6302; Dalton Trans. 2018, 47, 6274-6278; J. Organomet. Chem. 2019, 880, 108-115; ChemistrySelect 2020, 5, 9900-9907.
Synthesis and Applications of Inorganic Macrocycles & Cryptands
Synthesis of small and medium sized macrocycles containing acceptor atoms in their backbone
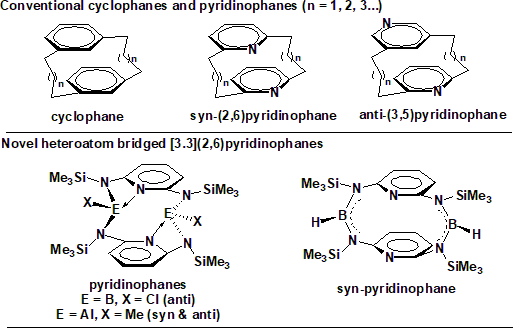
Synthesis of pyridinophanes and bicyclic pyridinophanes: Pyridinophanes are considered derivatives of cyclophanes with one or both of the benzene rings replaced by pyridine (see figure). Pyridinophanes have additional donor sites that can provide tuneable conformations compared to the conventional cyclophanes.
Synthesis of pyridinophanes and bicyclic pyridinophanes: Pyridinophanes are considered derivatives of cyclophanes with one or both of the benzene rings replaced by pyridine (see figure). Pyridinophanes have additional donor sites that can provide tuneable conformations compared to the conventional cyclophanes.
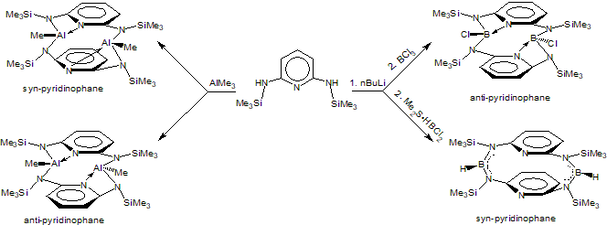
Conformationally rigid ‘‘N–E–N’’ heteroatomic bridged [3.3](2,6)pyridinophanes as the first examples of group 13 element containing pyridinophanes have been assembled using heteroatom N–E–N bridges (E = B, Al). The presence of B and Al as acceptor atoms in the bridges and their coordination with pyridine nitrogen has a very strong influence on the conformational rigidity of the pyridinophanes. The tetraazadibora pyridinophane with B-Cl miety adopts anti conformer whereas the B-H containing pyridinophane adopts syn (boat,boat) conformer. The Al analogues have been found to adopts both the syn and anti conformers.
For details see: Chem. Commun. 2018, 54, 8857-8860; Dalton. Trans. 2019, 48, 7442-7450; J. Organomet. Chem. 2019, 880, 108-115.
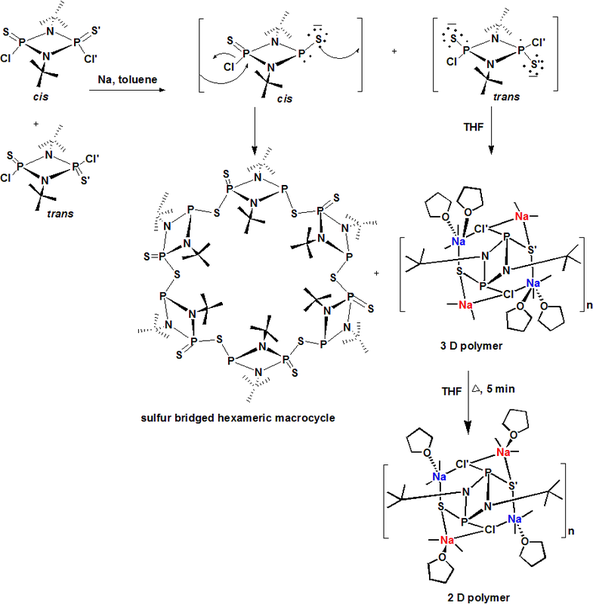
Inorganic Macrocycles and Cryptands: Synthesis of phosphazane based and boron-amide based organic-inorganic hybrid macrocycles and cryptands are major theme of this research area. We have successfully assembled a series of new pyridinophanes and cryptands. Study of host-guest complexation and use of cations, anions or neutral molecules as templates in assembling macrocycles and cryptands are important aspect of our work. A representative example shown below nicely explains how the disodium salt of the trans biradicaloid dianion, based on phosph(V)azane, forms 2D and 3D polymers whereas the cis form produces a hexameric sulfur bridged inorganic macrocycle.